
Choose a channel
Check out the different Progress in Mind content channels.

Progress in Mind
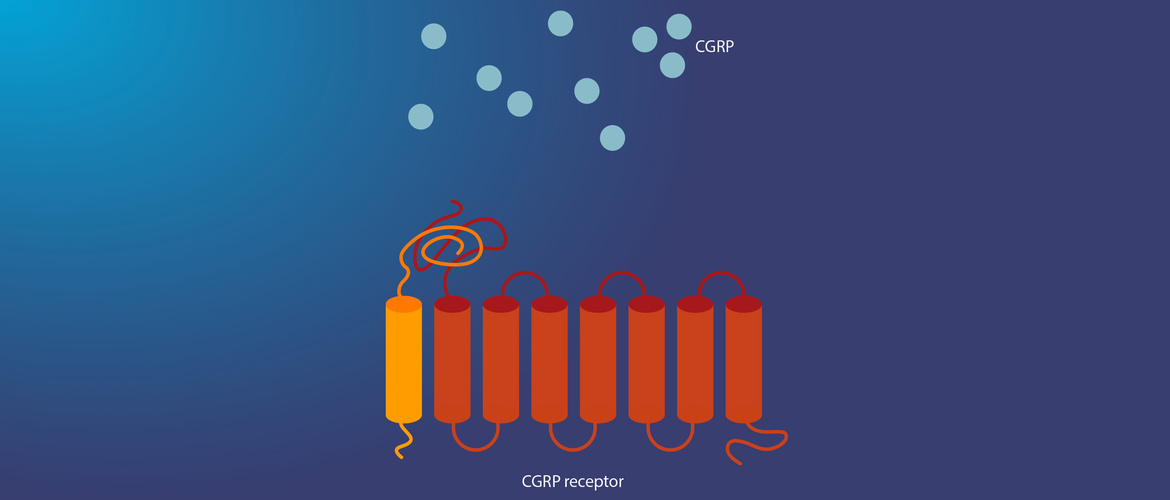
The involvement of calcitonin gene-related protein (CGRP) in the pathology of migraine has long been known, but it has taken a long time to develop treatments targeting the CGRP system. Recent advances have now renewed interest in CGRP-based therapies. Professor Uwe Reuter of Charité Universitätsmedizin, Berlin, Germany, gave us a glimpse of the future at the World Congress of Neurology 2019.
The involvement of CGRP in migraine was first proposed in 1986, only a few years after its discovery. Soon afterwards, a landmark study found significant release of CGRP during acute migraine and cluster headache attacks.1 Our understanding of the role of CGRP has since evolved alongside our understanding of migraine.
A promising start
CGRP is a short, 37 amino acid protein generated through alternative splicing and expressed throughout the body. It has functions in cerebrovascular regulation, as shown in a seminal study which demonstrated its vasodilator effect on pre-contracted arteries in vitro.2 Further observations in patients revealed that the levels of CGRP in blood increase during a migraine attack in the jugular vein located on the same side of the body as the migraine pain.3
New classes of migraine therapy have emerged targeting CGRP
During an episode, CGRP is released from trigeminal afferent neurons and binds to post-synaptic CGRP receptors (CGRP-Rs). The receptors are located either on the walls of meningeal blood vessels, where CGRP acts as a vasodilator, or on secondary neurons, where CGRP acts as a neurotransmitter.4 It is the neurotransmitter action in the trigeminal ganglion that is thought to contribute to migraine pain.
A new age
As a result of this knowledge, researchers began to consider that antagonism of the CGRP system could treat migraine. There is now renewed interest as three new classes of migraine therapy have emerged that aim to reduce the activity of CGRP:
New classes of therapy aim to reduce CGRP activity
These new classes target different aspects of migraine. The mAbs are aimed at treating chronic migraine, and the CGRP-R antagonists tend to be used for acute therapy of episodic migraine. This raises an interesting consideration when these medications are used in the clinic. If a patient is given a CGRP-R mAb as preventative therapy, will a small molecule CGRP antagonist then be effective as treatment for an acute migraine attack? There is no definitive answer to this question yet.
A different type of molecule
Professor Reuter focused on the mAb medications. A long half-life of around 30 days in the body allows an interval of 1 month between doses or longer.
The large size of the molecules means mAbs cannot cross the blood-brain barrier in clinically significant amounts.5 Although a patient feels their migraine pain in their brain, it appears that the trigeminal ganglion controls the sensory signal. Therefore their size does not affect their therapeutic action at the trigeminal ganglion. Crucially, the mAbs also do not cause raised levels of liver enzymes.
Our correspondent’s highlights from the symposium are meant as a fair representation of the scientific content presented. The views and opinions expressed on this page do not necessarily reflect those of Lundbeck.
1. Edvinsson L, et al. Nat Rev Neurol 2018;14(6):338-50.
2. McCulloch J, et al. Proc Natl Acad Sci U S A 1986;83(15):5731-5.
3. Schuster N and Rapoport A. Nat Rev Neurol 2016;12(11):635-50.
4. Doods H, et al. Trends Pharmacol Sci 2007;28(11):580-7.
5. Silberstein S, et al. Headache 2015;55(8)1171-82.